 Based on this, we can define two types of chemical reactions: exothermic and endothermic.Įxothermic reactions are those in which there is a release of heat and energy is given out to the surroundings. Δ ( PV) = change of Pressure x Volume of the systemĪs enthalpy is a state function and it is dependent on the changes between the initial and the final state. Knowing if the enthalpy of the system increases or decreases,during a chemical reaction is a crucial factor to understand if that reaction can happen.The change in the enthalpy of the system during a chemical reaction is defined as the change in its internal energy plus the change in the product of the pressure times the volume of the system: If this system is a chemical reaction, the change of heat is called enthalpy change. As we all know, the heat can go in or out of the system. EnthalpyĮnthalpy is the heat content of a system. Third law: The entropy of a perfect crystal is zero when the temperature of the crystal is equal to absolute zero (0 K). Second law: In an isolated system, natural processes are spontaneous when they lead to an increase in disorder, or entropy. First law: Energy is conserved it can be neither created nor destroyed. Thermodynamics is the study of the relationship between heat and work and we need to be familiar with the 3 important laws of thermodynamics, in order to understand this topic. 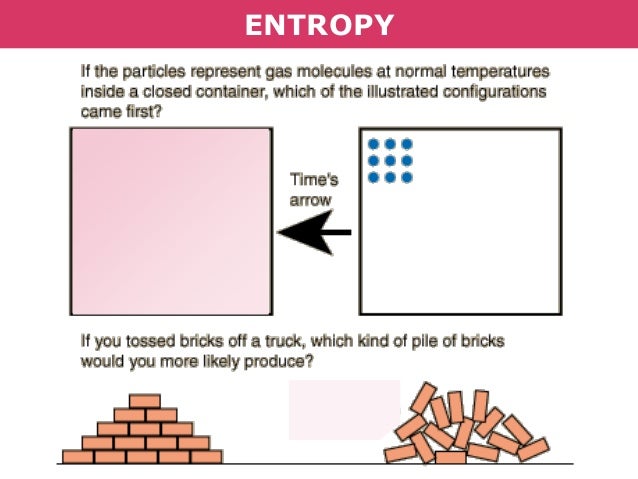
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
